In an extraordinary breakthrough by University of California, Irvine researchers, A nano wire based battery has shown promising results with more than 200 thousand recharge cycles. Post testing it for up to 200,000 cycles, they published their findings in American Chemical Society’s Energy Letters. The battery employs nano wires coated in a gel electrolyte to prevent from cracking. This could possibly revolutionize the purchase of a second battery. Imagine buying a battery for you laptop, and it doesn’t go out on you, ever.
Since nano wires are thousands of times thinner, these offer a larger surface area for transfer for storage and conduction of electrons. However these often grow brittle over large number of recharge cycles, and may break down over long use. The concept of using Nano-wires in batteries is not new, and has been for some time now. The research by UCI researchers has solved this problem. coating a gold nano-wire in a manganese dioxide shell and packing this assembly in a Plexiglas-like gel electrolyte. The experimental stages have been very promising, and the battery looks reliable.
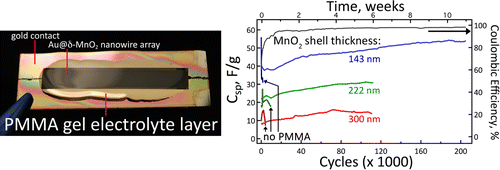
Image courtesy: APC Publications
UCI doctoral candidate, Mya Le Thai, the study leader, cycled the electrode up to 200,000 times over three months, with 94-96% efficiency.
The Battery Research Team Talks:
“Mya was playing around, and she coated this whole thing with a very thin gel layer and started to cycle it,” said Reginald Penner, chair of UCI’s chemistry department. “She discovered that just by using this gel, she could cycle it hundreds of thousands of times without losing any capacity.”
“That was crazy,” he added, “because these things typically die in dramatic fashion after 5,000 or 6,000 or 7,000 cycles at most.”
The researchers think the gel plasticizes the metal oxide in the battery and gives it flexibility, this prevents the nano-wires from cracking.
“The coated electrode holds its shape much better, making it a more reliable option,” Thai said. “This research proves that a nanowire-based battery electrode can have a long lifetime and that we can make these kinds of batteries a reality.”










